Technical Note 107: Basic Principles of SPRm Impedance
Electrochemical impedance spectroscopy (EIS) is a technique traditionally used in corrosion monitoring, coatings evaluation, batteries, and electrodeposition and material characterization. In recent years, this technique has gained momentum in biotechnology, …
Technical Note 106: SPR Microscopy vs Radioligand Binding Analysis
Radioligand binding assay has been used to measure the affinity of ligand binding to membrane protein receptors, including G-protein coupled receptor (GPCR). The first radioligand binding assay was performed in …
Technical Note 105: Introduction of SPR Microscopy
Surface Plasmon Resonance (SPR) is a widely used label-free detection technique for studying binding behavior of biomolecules. Since its commercialization in 1990’s [1], SPR has made vast advances in terms …
Technical Note 104: Concepts Behind EC-SPR
Voltametric technique in electrochemical (EC) detection is a very powerful analytical method that has been used for a wide range of applications. When combined with SPR technology, EC-SPR provides many …
Technical Note 103: Surface Plasmon Resonance v. Quartz Crystal Microbalance
Surface Plasmon Resonance (SPR) and Quartz Crystal Microbalance (QCM) are two powerful label-free detection techniques that are receiving increasing amounts of attention for many applications, ranging from biomedical, electrochemical, and surface studies to chemical- and biosensors. An often-asked question is: Which one is better or more suitable for my applications? This note will discuss the differences between the two techniques and compare some of the key performance parameters, such as sensitivity, response time, detection limit, accuracy, and multiplexity.
Technical Note 102: SPR Sensitivity and Detection Limit
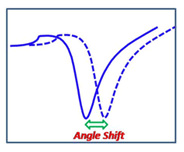
When selecting a Surface Plasmon Resonance (SPR) instrument, an obvious question to ask is: What is the sensitivity of the instrument? This seemingly simple question does not have a straightforward answer. The confusion is partially due to the fact there is simply no perfect way to define the sensitivity, and also partially due to the inaccurate use of this term by some vendors. This technical note describes some of the most commonly used terms for defining SPR sensitivity. The goal is to provide SPR users with a guideline to determine if a particular definition is useful for his/her application, and to compare different instruments in a meaningful way.
Technical Note 101: Principle of SPR Detection: Intensity profile and shift of the SPR angle
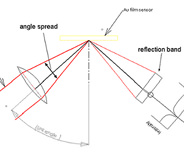
Principle of SPR Detection: Intensity profile and shift of the SPR angleWhen a light beam impinges onto a metal film at a specific (resonance) angle, the surface plasmons are set …
