Fixation of cells is a convenient and routinely used method by many labs to study cells. However, fixation may alter certain types of protein receptors, thus affecting their binding kinetics. For these receptors, direct measurement of the binding kinetics on live cells becomes crucial.
This application note describes the application of SPRm 200 to measuring the binding between lectin and glycoprotein on live cells in a cell medium. Glycoproteins are membrane proteins that play an important role in cell recognition and communications. (1,2) The binding of wheat germ agglutinin (WGA, 36k MW), a lectin that recognize N-acetylglucosamine (GlcNAc), was studied on live CHO cells seeded on a SPRM chip. The measurement was performed using a CO2 independent cell culture medium with WGA concentrations from 0.8 nM to 200 nM.

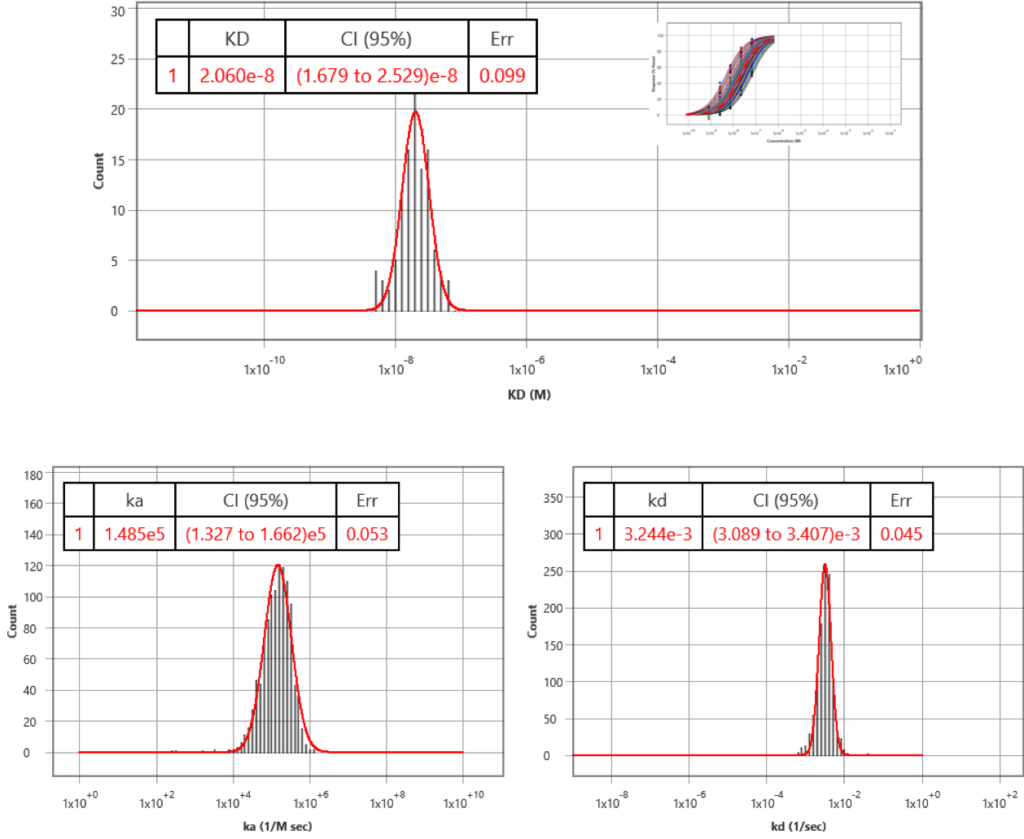
Kinetic data of few hundred Regions of Interest (ROIs) were analyzed, and kon, koff and KD obtained by fitting the data with a simple kinetic model were 1.5 x 105 M-1 s-1 (95% CI 1.3 – 1.7), 3.2 x10-3 s-1 (95% CI 3.1 – 3.4) and 22.1 nM (95% CI 20.2 – 24.1), respectively. In addition to the kinetic analysis, the equilibrium analysis was also performed, with KD= 20.6 nM (95% CI 16.8 – 25.3), which is consistent with the kinetic analysis data.
This example demonstrates the application of SPRm for studying membrane protein binding kinetics on live cells and in a complex medium. Measuring cell binding responses in live conditions further expands the benefits of using SPR microscopy technology especially to research of antibodies binding to cell membranes proteins.
We thank Novartis Pharma AG Switzerland for the useful discussions.
Author: Nguyen Ly | Biosensing Instrument | Published Jan 4, 2025
DOWNLOAD PDF
Download a PDF of Application Note 135: Measuring Binding Kinetics in Cell Medium with SPRM
- Dell, A et al, Science, 291, 2351-2356, 2001
- Durand, G et al, Clinical Chemistry, 46, 795–805, 2000
