Entry level surface plasmon resonance (SPR) tools with dual channels have only one measurement point since the other channel is always used as a reference.1 Therefore, increasing efforts have been made to come up with SPR systems with multiple channels with more flexibility in terms of SPR assay development. This application note focuses on the benefits of the 5 channel SPR instrument (BI-4500), which offers several advantages over dual-channel SPR. Firstly, the ability to simultaneously and quantitatively detect multiple analytes with increased throughput. Secondly, comprehensive analysis of complex samples like biological fluids can be done at ease with faster response times.2 Thirdly, the serial format of multiple channels can be used either to increase the throughput for measurements of a single analyte or to increase assay diversity by measuring multiple analyte-ligand pairs in a single run. Fourth, is the ability to analyze multiple samples on different channels allowing direct comparison between them while controlling experimental variations like non-specific binding, temperature fluctuations resulting in accurate readings compared to dual-channel conditions.
Small molecule immunosensing using SPR is more challenging. However, new developments are increasingly meeting these challenges to offer highly sensitive small molecule immunosensor technologies through careful consideration of sensor interface design and signal enhancement. Depending on the experimental design, multichannel SPR can offer enhanced sensitivity by allowing precise control over the interaction conditions in each channel.3 To evaluate the sensitivity of the multichannel instrument, binding interactions between four different small molecule inhibitors with the surface immobilized enzyme carbonic anhydrase II was studied using a single dextran chip (Figure 1). The enzyme system was used to compare the binding thermodynamics and kinetics of four sulfonamide inhibitors (Furosemide, Dansylamide, Acetazolamide and Sulfanilamide) with varied masses from 172 to 330 Daltons.

Figure 1: Schematic of the SPR sensor for detecting the interaction of Acetazolamide, Furosemide, Sulfanilamide and Dansylamide with CAII preimmobilized onto channels CH 1-5.
All the compounds were assayed over a 1000-fold concentration range using a 2-fold dilution series. A visual comparison of the four different kinetic binding constants of the small molecule inhibitors was made by plotting the association versus the dissociation rates for each compound (Figure 2).
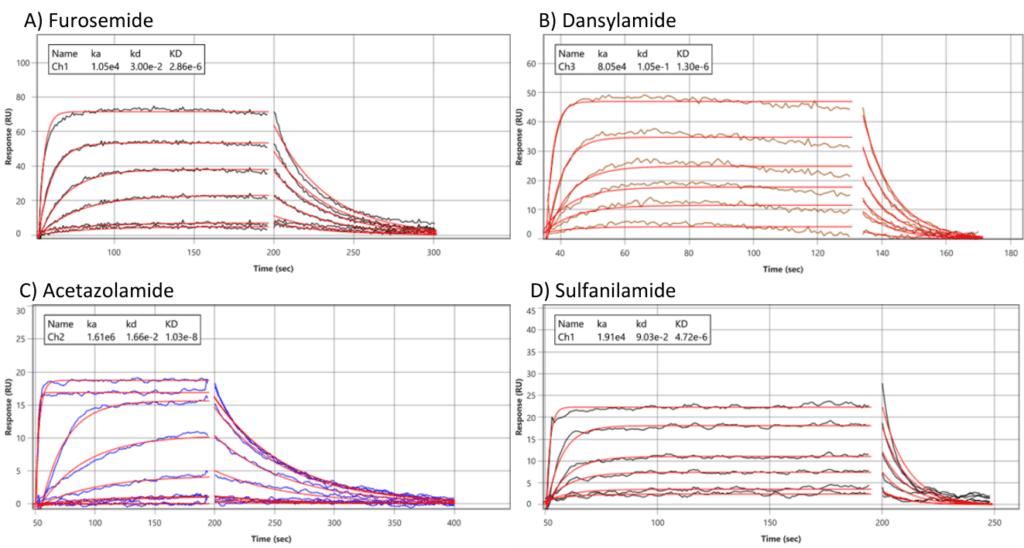
Figure 2: Sensorgrams for sulfonamide-based inhibitors interacting with a CAII surface. The highest concentration for each compound was as follows: A) Furosemide (40 μM) B) Dansylamide (80 μM) C) Acetazolamide (10 μM) D) Sulfanilamide (500 μM) Each compound was injected over a 1000-fold concentration range using a 2-fold dilution series. The entire data set was globally fit to a 1:1 interaction model as shown by red lines.
The reaction and reference channels are on the same chip, which makes it easier to resolve the dissociation kinetics for very rapidly dissociating systems. The instrument also showed the ability to monitor multiple interactions in real-time with kinetic details, providing insights into the different dynamics of four analytes with a ~10,000-fold difference in affinity to the same ligand.
| Compounds | Molecular weight (Da) | ka (M-1 s-1) | kd (s-1) | KD |
| Furosemide | 330.7 | 1.05 e4 | 3.00 e-2 | 2.8 uM |
| Dansylamide | 250.3 | 8.05 e4 | 1.05 e-1 | 1.3 uM |
| Acetazolamide | 222.2 | 1.61 e6 | 1.66 e-2 | 10 uM |
| Sulfanilamide | 172.2 | 1.91 e4 | 9.03 e-2 | 4.7 uM |
Table 1: Kinetic values of small molecule binding to CAII.
Also, the rate constants determined from this high-resolution analysis (Table 1) are consistent with the values determined from our previous studies.4
The capability to detect and quantify multiple analytes in a single experiment is particularly useful for studying complex biological interactions or analyzing mixtures of molecules. Zhou et al used BI-4500 to simultaneously functionalize a five-channel sensor for labeling free diagnostics. 2 To achieve this, SARS-CoV-2 spike (S1) protein and angiotensin-converting enzyme 2 (ACE2) were immobilized into separate channels on the same tris-nitrilotriacetic acid (tris-NTA) chip to simultaneously study their interaction with anti-S1 antibody and viral particles in serum samples (Figure 2).The optimal density of probe molecules in each fluidic channel was precisely controlled through the closure and opening of the specific channels. In addition to being sensitive, accurate, and high throughput of the mentioned species in complex serum samples, this sensor can also be used for evaluating the neutralizing ability of a given antibody and for differentiating free viral particles from their neutralized counterparts.
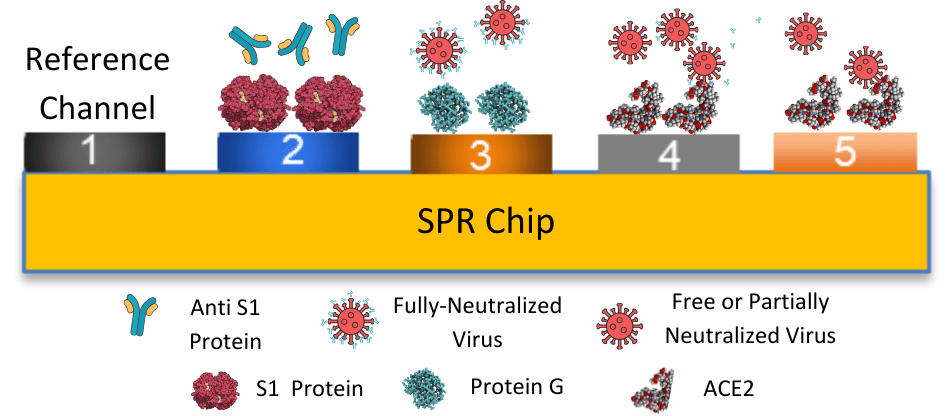
Figure 3: Schematic of the SPR sensor for detecting free anti-S1 antibody, viral particles fully or partially neutralized viral particles, with His-tagged S1 protein pre-immobilized onto channel CH 2, His-tagged protein G onto CH 3, and His-tagged ACE2 onto CH 4 and CH 5.
This technique is versatile as serum samples with antibodies and virus levels corresponding to infections/immunities in different types of sera can be assessed in a single test, facilitating rapid assessment of immunity, disease status, and vaccine efficacy.5
The flexibility to customize each channel with different immobilized ligands or detection conditions to study a variety of interactions simultaneously can be done at ease using the multi-channel SPR systems. These advantages make this sensor technology an ideal tool to support secondary screening in drug discovery. Additionally, detecting multiple interactions on a single chip enables comparison of results with faster response times, lowering the costs. It also enhances sensing capabilities by allowing for more comprehensive analysis of complex samples by monitoring multiple interactions at once. Also, the high sensitivity delivers high quality binding response for low immobilization and small molecules drug research using multichannel SPR.
Author: Nguyen Ly and Miyuki Thirumurthy | Biosensing Instrument | Published March 17th, 2025
DOWNLOAD PDF
Download a PDF of Application Note 164: Simultaneous Analysis of Binding Interactions using Multi-Channel SPR
- Singh, Pranveer.,B: Chemical 229 (2016): 110-130.
- Dong, Tianbao, Chaowei Han, Meng Jiang, Tiantian Zhang, Qing Kang, Pengcheng Wang, and Feimeng Zhou, ACS sensors 7, no. 11 (2022): 3560-3570.
- Mitchell, J. (2010),Sensors, 10(8), 7323-7346.
- Myszka, David G, Analytical biochemistry 329, no. 2 (2004): 316-323.
- Rocha, V. P. C., Quadros, H. C., Fernandes, A. M. S., Gonçalves, L. P., Badaró, R. J. D. S., Soares, M. B. P., & Machado, B. A. S. (2023), Viruses, 15(7), 1504.
