Streptavidin (SA) sensor chips are commonly used for kinetic interactions of biotinylated ligands. Streptavidin is a tetrameric molecule with an extremely high affinity towards biotin and, because of its binding capacity, reproducibility and chemical resistance,streptavidin is widely used for bioconjugation. SA chips facilitates detection of small molecules, as well as the use of harsh regeneration protocols with retained cycle-to-cycle reproducibility.[1]
In this note, the assay was run with streptavidin immobilized onto the SPR sensor chip to capture biotinylated peptides (Figure 1). To measure the dissociation equilibrium constant between the molecules, a five-channel SPR (BI-4500) and a streptavidin sensor chip from Biosensing Instrument were used.
T10-39 (FFDRYDSARDPGRLL), a member of the library of mRNA-displayed peptide, was found to bind to human α-thrombin. Human α-thrombin is a key enzyme in blood coagulation modulating as a procoagulant as well as an anticoagulant.[2] Natural inhibitors of human α-thrombin such as heparin, hirudin [3], and hirugen [4] serve as anticoagulants in a variety of clinical applications. These examples show that the human α-thrombin is a protein recognized and affected by a wide variety of protein motifs. [5]
Biotinylated Th10-39 was immobilized onto four of the five fluidic channels, which were subsequently exposed to various concentrations of human α-thrombin (0.63, 2.50, 5, 10, 15, 25 nM) at a flow rate of 60 μL/min (Figure 2). Background, collected from the fifth channel (i.e. a Th10-39-free channel that was also exposed to human α-thrombin), was subtracted from data collected from the first four channels. PBS was used as running buffer and regeneration injection of 20 mM NaOH followed each binding interaction. The overlaid sensorgrams (black line curves) fitted with a 1:1 kinetic model (red line curves) (Figure 2). The association rate constant (ka) is found to be 3.5 × 106 M-1s-1, dissociation constant rate (kd) of 3.9 × 10-2 s-1 and equilibrium dissociation constant (KD) of 10.9 nM.
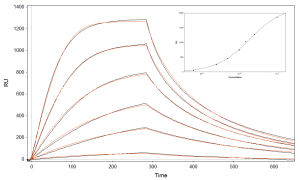
FIG. 2 Overlaid experimental and simulated sensorgrams corresponding to the interactions between the immobilized biotinylated Th10-39 and human α-thrombin at six different concentrations. (Inset) A plot of the maximum responses plotted against the concentrations of human α-thrombin along with the Langmuir isotherm.
In summary, by using the SPR technique and SA chips, it is straightforward to obtain the kinetic and thermodynamic parameters related to biotinylated ligands.
Author: Nguyen Ly | Biosensing Instrument | Published Jan 4, 2025
DOWNLOAD PDF
Download a PDF of Application Note 123: Surface Plasmon Resonance Measurement of Protein-Peptide Interaction Using Streptavidin Sensor Chip
- https://www.sprpages.nl/sensor-chips-intro/biacore-sensor-chips/sa
- Stubbs, M.T and Bode, W., Trends Biochem. Sci., 1995, 20, 23-28
- Stone, SR and Hofsteenge, J., Biochemistry, 1986, 25, 4622-4628
- Skrzypczak-Jankun, E., et al, J. Mol. Biol., 1991, 221, 1379-1393
- Raffler, N. et al, Chemistry & Biology, 2003, 10, 69-79