SPR Sensitivity and Detection Limit
When selecting a Surface Plasmon Resonance (SPR) instrument, an obvious question to ask is: What is the sensitivity of the instrument? This seemingly simple question does not have a straightforward answer. The confusion is partially due to the fact there is simply no perfect way to define the sensitivity, and also partially due to the inaccurate use of this term by some vendors. This technical note describes some of the most commonly used terms for defining SPR sensitivity. The goal is to provide SPR users with a guideline to determine if a particular definition is useful for his/her application, and to compare different instruments in a meaningful way.
Angular Sensitivity
The most popular SPR detection scheme uses the so-called Kretchmann configuration and measures the angle of incident light at which surface plasmon resonance takes place (see Figure 1 below). The shift of the resonance angle provides a sensitive measurement of a molecular binding event onto the sensor surface or a change in the index refraction of the fluid medium near the sensor surface. For this reason, the minimum detectable angular shift, in unit of degree, may be used to describe the sensitivity. However, an SPR instrument with the best angular sensitivity does not always mean that it has the best sensitivity in terms of detecting molecular binding, which is referred to as surface sensitivity.
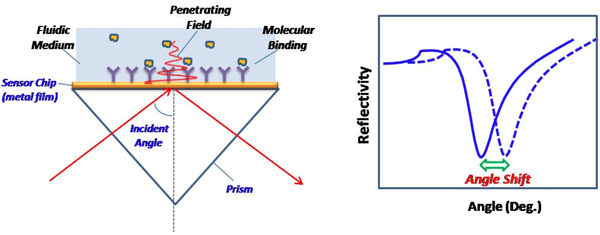
FIG. 1 Left: A typical SPR setup. An incident light is directed onto a SPR sensor chip via a prism, and the reflected beam is detected via a photodetector or imager. At an appropriate angle (resonance angle), the incident light excites the surface plasmons in the sensor chip (metal film) and the intensity of the reflected light drops to a minimum. The electromagnetic field created by SPR penetrates the fluidic medium and probes molecular binding processes taking place on the surface and the refractive index changes in the fluidic medium. Right: Reflectivity vs. incident angle plot shows a sharp drop in the reflection intensity due to SPR, also referred to as the SPR “dip”. The angular position of the dip is often measured and used to define SPR sensitivity.
Dependence upon Prism Material
It is worth noting that the resonance angle depends not only on molecular binding and the index of refraction of the fluid medium, but also on the index of refraction of the prism and the dielectric constant of the metal film, as well as on the wavelength of light used to excite the surface plasmons. When comparing the sensitivities of different instruments in terms of degree angles, one should be aware of the prism material and the metal film, as well as the wavelength of light.
Example 1: If the prism is made of BK7 (n=1.515) glass and the wavelength of incident light is 635 nm, then the angular shift due to a protein binding layer (n= 1.5) of 3 nm on a gold sensor chip is 0.75 deg. If keeping everything the same, except replacing the BK7 glass prism with a SF10 glass (n=1.723) prism, then the same protein binding layer leads to an angular shift of 0.35 deg (a weaker response). If two instruments report the same angular shift, the one using BK7 prism is actually more sensitive in terms of measuring molecular binding.
Dependence upon Wavelength
The penetration length of the evanescent field created by SPR into the fluid medium increases with the wavelength. Longer wavelengths (e.g., near infrared) have the “seeming” advantage of being able to probe further beyond the sensor surface; however, this results in a significant loss of surface sensitivity.
Example 2 Two SPR instruments both use BK7 glass prisms, gold sensor chips, and have similar angular sensitivity, but one uses 635 nm light and the other uses 890 nm light. For a protein binding layer of 3 nm, the first instrument produces 0.75 deg angular shift, but the second instrument leads to only 0.2 deg angular shift (a much weaker response). For two instruments that have similar angular shifts, the one using 635 nm light is actually more sensitive in terms of measuring molecular binding. Although longer wavelengths allow for slightly deeper detection into the solution bulk, this results in a significantly lower sensitivity for measuring molecular binding on the sensor surface.
Example 3 Two SPR instruments claim to have similar sensitivities since they have matching values for angular sensitivity. However, one instrument uses a BK7 glass prism and 635 nm light, while the other instrument uses a SF10 glass prism and 890 nm light. For a protein binding layer of 3 nm, the first instrument produces 0.75 deg angular shift. Not surprisingly though, the second instrument results in a much weaker response of 0.15 deg angular shift. For two instruments that have similar angular sensitivity, the one using 635 nm light and BK7 glass is actually 5 times more sensitive in terms of measuring molecular binding.
Relative Index of Refraction Unit
Another quantity often used to describe SPR sensitivity is the relative change in the index of refraction of the fluid medium, known as RIU. Unlike angular shift, the unit RIU is more relevant to applications that demand an accurate measurement of the index of refraction of a bulk fluid. As a result, RIU may not be the most convenient unit for applications that aim to study molecular binding events. A relationship between RIU and angular shift in degrees is possible if one knows the exact instrumental conditions (e.g., wavelength of incident light and material of prism glass). Just like with angular shift, a SPR instrument that has the best sensitivity in terms of RIU does not always mean that it has the best sensitivity in terms of detecting molecular binding.
Example 4 If an SPR instrument uses a BK7 glass prism, gold sensor chip, and 635 nm incident light, then a 0.010 RIU change in aqueous buffer solution results in ~1.55 deg angular shift. However, this conversion between RIU and angular shift is not universal, as it depends upon the instrumental conditions (e.g., wavelength of incident light and prism material). By increasing the wavelength to 890 nm while keeping all the other experimental parameters the same, a new relation is observed in which a 0.010 RIU change in aqueous buffer solution results in a smaller ~0.99 deg angular shift. Additionally, if both the wavelength is increased to 890 nm and the prism glass is changed to SF10, then a 0.010 RIU change in aqueous buffer solution results in ~0.61 deg angular shift. Thus, the comparison of sensitivity between units of degree angular shift and RIU requires careful consideration of instrumental conditions.
Example 5 How do the following SPR instruments compare? The first instrument has a BK7 glass prism with 635 nm light and a sensitivity of 0.1 mDeg. The second instrument has a SF10 glass prism with 890 nm light and a sensitivity of 1 µRIU. From Example 4, we learned that 0.010 RIU corresponds to 0.61 deg for this configuration. Using this relationship, 1 µRIU corresponds to a sensitivity of 0.06 mDeg. Does this mean that the second instrument is more sensitive than the first? No. Remember that angular sensitivities alone do not tell the complete story. We must determine the surface binding sensitivity to make the fairest comparison. From Example 3, we learned that the configuration of the second instrument is 5 times less sensitive than the configuration of the first instrument. Thus, a sensitivity of 0.06 mDeg actually corresponds to an equivalent surface binding sensitivity of 0.3 mDeg. As a result, the first instrument is experimentally more sensitive than the second.
Surface Coverage
If one is interested in using SPR to detect molecular binding taking place on a sensor surface, then surface coverage in terms of mass, e.g., pg/mm², is an appropriate way to define sensitivity. The unit RU (termed Resonance Unit or Response Unit) is defined as 1 RU = 1 pg/mm², and is also often used to determine surface coverage.
However, this description cannot be used ubiquitously. For instance, SPR measures the optical polarizability, size, and density of the molecules bound to the surface, which are related to but different from an SPR measurement in terms of mass per unit surface area. The polarizability depends on the wavelength of light, especially if the wavelength is close to the optical absorption band of the molecule (e.g. chromophores, UV-vis labels, etc.). Since most proteins have similar polarizabilities, the SPR signal may be considered approximately proportional to the coverage of molecules bound to the sensor surface, and pg/mm² is a useful way to quantify SPR sensitivity.
Example 6 A monolayer of Cytochrome c leads to an angular shift of ~0.5 Deg. The corresponding mass coverage is ~3000 pg/mm². For an angular sensitivity of 0.1 mDeg, the corresponding mass sensitivity is 0.6 pg/mm² or 0.6 RU.
Molar Concentration
Some vendors provide sensitivity in terms of lowest detectable molar concentration. This is a convenient and attractive measure of SPR instrument sensitivity. However, the reality is that a highly sensitive instrument cannot faithfully guarantee the detection of an extremely low analyte concentration. This is because sensitivity and detection level are two different (although related) analytical “figures of merit” [1] which are often mistakenly mixed. The lowest detectable molar concentration depends upon several significant experimental factors such as the molecular weight, optical property, and binding affinity of the analyte, as well as the surface coverage of the capture molecules. Background noise also plays a key role in determining the lowest detection level (more about this later). Molecules with large molecular weight and polarizability are easier to detect than those with small molecular weight and polarizability. A high affinity and surface coverage of the capture molecules also facilitate the detection of analyte molecules per given concentration. Additional factors that influence the lowest detectable molar concentration include sensor chip preparation (e.g., the thickness of the modifier layer and its refractive index), temperature, and buffer solution performance. Moreover, numerous experimental strategies exist that can amplify SPR binding responses (e.g., labels, competitive binding assays, enzymatic reactions, etc.). As a result, SPR sensitivity in terms of lowest detectable molar concentration can be misleading, and very unforgiving to beginning SPR users.
Example 7 A sensor chip is functionalized with 5 x 10-16 mol/mm² anti-PNA (peanut agglutinin). PNA molecular weight is about 100 kDa, and the PNA-anti PNA equilibrium dissociation constant, K, is about 20 nM. For a SPR instrument with a sensitivity of 0.1 mDeg, or corresponding mass sensitivity of ~0.6 pg/mm², the minimum detectable concentration will be ~0.5 nM at equilibrium. Clearly, if factors such as the surface coverage and equilibrium dissociation constant were different, then the minimum detectable concentration would also change. Thus, the evaluation of sensitivity in terms of analyte concentration should be carefully considered, keeping in mind that chip and experimental conditions play critical roles.
Determination of detection levels
The definition of “lowest detectable level” is often not clearly spelled out. The lowest detection level is largely governed by the background noise. Some choose the peak-to-peak value of the noise in the SPR signal, while others use root-mean-square or standard deviation. In analytical chemistry, an often-used definition of detection limit is three times the standard deviation of the background (blank) noise. Second, the noise of a measured physical quantity usually occurs at various time scales, so SPR sensitivity should be given together with the time scale of the measurement. Filters, such as time averaging and smoothening of data, can remove certain noises and improve the sensitivity and detection level. This practice tends to slow down the response time. One must also make sure that the response time is fast enough for an application when choosing an instrument. Third, the noise level may be influenced by electronic amplification (or gain control). A higher gain may improve signal to noise ratio, but this usually affects the dynamic range (detection range) of the instrument. Finally, when compared with imaging SPR or other pixel-based detectors, the sensitivity depends on how many pixels the SPR signal is averaged over and for how long. More pixels and more time lead to better sensitivity, but it may sacrifice spatial resolution and response time.
Nguyen Ly and NJ Tao | Biosensing Instrument | Published March 1, 2008
Reference:
[1] Skoog, D. A.; Holler, F. J.; Crouch, S. R. Principles of Instrumental Analysis, 6th ed.; Thomson Higher Education, Belmont, CA, 2007.
