Some biomolecules such as enzymes can exhibit much greater biological activities at physiological temperature (37.5 °C) or higher (e.g., DNA polymerase). The focus of this study is to screen for potential inhibitors of β-site amyloid precursor protein cleaving enzyme 1 (BACE1). BACE1 is an enzyme that cleaves the transmembrane amyloid precursor protein to produce the amyloid beta (Aβ) peptides,[1] whose subsequent aggregation produces neurotoxic aggregates that initiate neuronal cell death and the evolvement of Alzheimer’s disease.[2]
The cleaving activity of BACE1 enzyme at room temperature is very weak, but is strong at 37 °C. Peptides were immobilized onto a CM Dextran surface for screening. Confirmation testing of the peptide substrate was accomplished by exposures to an Aβ-specific antibody, which only binds to intact (i.e., uncleaved) peptides.[3] Figure 1 shows confirmation testing of an Aβ-specific antibody to the peptide substrate before (black line) and after (blue line) exposure to BACE1 cleaving enzyme at an elevated 37 °C. The black line shows a strong binding interaction response, indicating the presence of uncleaved or intact peptides. For faster screening purposes, the peptide surface could be regenerated for retesting many times with 20 mM NaOH solution (denoted by the orange arrow, which causes the signal to return back to the original baseline). In contrast, the blue line shows significantly diminished antibody binding, indicating that the peptides were cleaved by the prior exposure to BACE1 enzyme at elevated 37 °C.
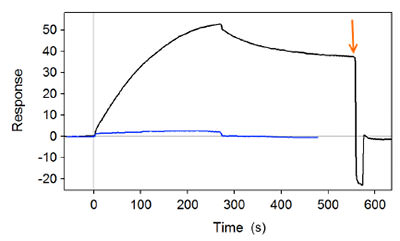
FIG. 1 SPR sensorgram showing the binding response of Aβ-specific antibody to the peptide substrate before (black line) and after (blue line) exposure to BACE1 cleaving enzyme at 37 °C.
Exposure of the peptide substrate to BACE1 enzyme at 25 °C produced curves similar to the black line, suggesting little cleavage of the peptide substrate is attained at a temperatures not optimal for the enzymatic activity. The precision temperature control feature of the BI-4000 SPR instrument was used to regulate the substrate and sample temperatures. This application note demonstrates a simple example where temperature control is an important feature for SPR measurements.
Author: Nguyen Ly | Biosensing Instrument | Published May 4, 2020
DOWNLOAD PDF
Download a PDF of Application Note 116: Temperature Dependence of Enzymatic Cleaving Activity
- Sinha et al. Nature 1999, 402, 537
- Hardy and Selkoe, Science 2002, 297, 353
- X. Yi et al., Anal Chem 2013, 85, 3660-3666
